Non-invasive methods for diagnosis of liver fibrosis: a systematic approach
Métodos não invasivos para o diagnóstico de fibrose hepática: uma abordagem sistemática
Graciele Gonçalves Santos1
Alexandre dos Santos Pyrrho1
1Universidade Federal do Rio de Janeiro (UFRJ) – Rio de Janeiro-RJ, Brasil.
Universidade Federal do Rio de Janeiro (UFRJ) – Rio de Janeiro-RJ, Brasil.
Conflito de interesses: não há conflito de interesses
Recebido em 24/05/2019
Artigo aprovado em 08/08/2019
DOI: 10.21877/2448-3877.201900852
INTRODUÇÃO
Liver fibrosis is a common consequence of several chronic liver diseases. In a first instant, through hepatic injury, an inflammatory process occurs. However, the initial damage is not responsible for causing the injuries themselves. From the moment the injury begins, an activation of cells of the immune system happens and then the release of pro-inflammatory cytokines, which culminate in the injury of liver tissue. Inflammatory mediators and other potentially toxic components accumulate leading to the generation of reactive oxygen species, which expose hepatocytes to oxidative stress, causing lipid peroxidation and DNA damage. While these events occur, several hepatic cells enter an “activated” state, resulting in increased synthesis and deposition of extracellular matrix components. The deposition is characteristic of liver fibrosis, which, in a large scale, could lead to a progression for cirrhosis, considered an irreversible condition wherein the liver architecture is altered and impaired.
One of the main cells in the activation process are the hepatic stellate cells (HSC), whereby their role as vitamin A storage cells change to become like myofibroblasts – contractile, proliferative and pro-fibrogenic, according to Taibi et al.(1) After HSCs activation, cytokines are secreted in an autocrine manner, generating signals that maintain their activation, survival and consequent extracellular matrix deposition. According to Ichikawa et al.,(2) a cycle is established in which the inflammatory and pro-fibrogenic stimulus contributes to maintain the fibrotic state of the liver.
Material and methods
Validation articles have been searched in four databases (Medline, Cochrane, LILACS and SciElo). The terms used were: (liver fibrosis) AND (non-invasive diagnosis). All the articles were recovered and succeed to the inclusion and exclusion methods. For this, titles, summaries and materials and methods were read.
Inclusion methods
- Validation articles for diagnostic methods;
- Validation of laboratory methods compared to standard gold diagnosis (liver biopsy);
- Reading of histopathological laminas performed by at least two pathologists who did not have information on the clinical status of the patients and chronic liver diseases present;
- Time interval between biopsy and blood tests of a maximum of six months, regardless of which was performed first.
Exclusion methods
- Works wherein non-invasive diagnostic methods are imaging tests, instead of the gold standard (liver biopsy);
- Languages other than English, Spanish or Portuguese;
- Articles that avoided the theme and/or focused on the treatment.
Therefore, the articles that matched all methods of inclusion and none of the methods of exclusion were completely read for further classification.
Results
The result of the search in the databases led us to a first screening of 139 articles. The subsequent selection and sorting stages are represented in Figure 1.
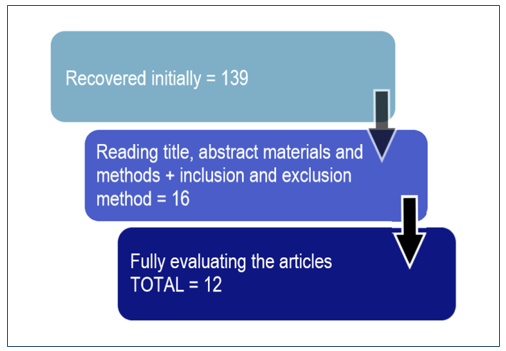
Figura 1. Fluxograma das etapas da triagem realizada para obtenção dos trabalhos.
About 10% of the retrieved articles could be used after screening by applying the inclusion and exclusion methods. It might have happened due to the repetition of several papers in the referred databases and many articles out of the topic and out of the kind of study of interest to this review. Selection of articles included studies ranging from 2005 to 2017.
Subsequently to the complete reading of the articles, some were excluded because they showed only biochemical markers associated with histopathological parameters, case of which would require a biopsy for the method, diverging to the proposal of this work (using non-invasive techniques). In other hand, some studies combined biochemical markers with imaging tests, not being comprised in the focus of this work, despite the non-invasive nature.
To complement the understanding of the techniques, its groundings, and pathophysiology of liver fibrosis, review articles have been searched in Medline database, prioritizing the most recent works that did not diverge from the purpose of the present theme.
Literature data
Several laboratory methods for the diagnosis and staging of liver fibrosis are presented below.
Serum homocysteine
Homocysteine is an essential amino acid derived from the metabolism of methionine. The reasoning for evaluation of this analyte during non-alcoholic fatty liver disease (NAFLD) course is that mild hyperhomocysteinemia is an important predictor of cardiovascular disease along with insulin resistance. It is known that insulin resistance plays an important role in the progression of NAFLD to non-alcoholic steatohepatitis (NASH), hence it has been questioned if homocysteine levels would also be altered in transition from NASH to fibrosis.
A study correlating their serum levels with the progression of fibrosis has been performed by Polyzos et al.(3) Here, the validation of the test was done in patients with NAFLD. It is known that NAFLD is composed of a spectrum ranging from simple hepatic steatosis to NASH, wherein inflammation and maybe progression to fibrosis, cirrhosis and even hepatocellular carcinoma might happen.
The tests were performed in 31 patients, 15 with NAFLD and 16 with NASH. For the control group, 31 people participated, with similar parameters (sex, age and body mass index). Serum homocysteine levels were lower in patients with NASH (mmol/L 12.3 ± 2.5) compared to individuals who had NAFLD (mmol/L 14.7 ± 2.1). Furthermore, comparisons between homocysteine levels were shown to be related to hepatic fibrosis stages, in an inverse manner. Table 1 lists levels of fibrosis correlated with serum homocysteine levels.
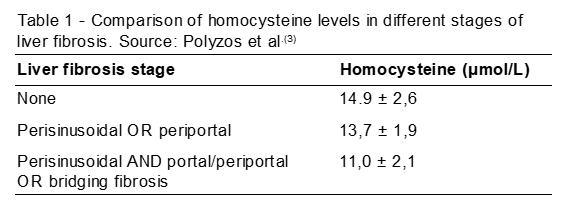
In addition to fibrosis, other parameters correlated with homocysteine levels (decreasing their levels according to disease progression), such as portal inflammation and steatosis location.
Homocysteine is an intermediate component in methionine metabolism and can follow two pathways: remethylated (a process requiring folate and vitamin B12), returning to the methionine state or irreversibly degraded to cysteine via the process of transulfuration (a process requiring vitamin B6).
Two reasons may explain why low homocysteine levels contribute to progression of NAFLD to NASH, and, consequently, fibrosis. The first one is the modulation of methyl balance: the reduction of homocysteine levels culminate in the decrease of S-adenosylmethionine, which in turn is necessary for exporting of VLDL from the liver. With the reduction of methionine (and consequent decrease in homocysteine), this process of exporting VLDL is decreased, increasing the hepatic accumulation of lipids. The second reason involves the enzyme cystathionine beta synthase, which catalyzes the condensation of homocysteine with serine to cystathionine, which in turn is hydrolyzed to cysteine, that could be used for the synthesis of glutathione. Low levels of glutathione promote the progression of liver fibrosis, because it protects liver cells from damage caused by oxidative stress. Therefore, measurement of serum homocysteine levels may be a good ally for the diagnosis of liver fibrosis in NAFLD cases.
Hyaluronic acid (HA) and vascular endothelial growth factor (VEGF)
Before understanding the reason for choosing these markers, it is necessary to emphasize some processes that occur in liver fibrosis. Its development is characterized by excessive deposition of extracellular matrix components, including various types of collagens, proteoglycans, structural glycoproteins and hyaluronic acid (HA). Such excess of extracellular matrix in the space of Disse causes a reduction in blood flow, affecting adjacent hepatocytes and influencing the clearance ability of these and other hepatic cells. Over time, a distortion of liver architecture comes up, forming fibrotic scars and regeneration nodules, which leads to hepatocellular dysfunction and increases intrahepatic resistance to blood flow. Additionally, during fibrosis, liver sinusoidal endothelial cells undergo transformations – they lose fenestrations. Thus, Hypoxia ends up being one of the consequences. This mechanism requires the participation of proinflammatory cytokines and growth factors, including vascular endothelial growth factor (VEGF). Furthermore, the increase of HA levels reflects in the capillary levels of liver endothelial sinusoidal cells, since it acts to promote the formation of endothelial cells and induce the expression of angiogenic factors, such as intercellular adhesion molecule 1 (ICAM-1), vascular cell adhesion protein 1 (VCAM-1) and matrix metalloproteinase 2 (MMP-2). Therefore, HA and VEGF may act as fibrogenic and angiogenic factors.
It is known that angiogenesis is essential for organ growth and repair after liver disease. The disturbance of balance between liver fibrogenesis and angiogenesis can lead to several diseases, including the onset of cancer. It is known that chronic hepatitis C is one of the main causes of the development of hepatic angiogenesis.
The study by Toson et al.(4) was validated in patients with chronic hepatitis C. The area under the curve (AUC) of HA to differentiate patients with significant fibrosis from those with non-significant fibrosis was 0.941 and of VEGF was 0.911. HA showed specificity of 94% and sensitivity of 85%, positive predictive value of 92% and negative predictive value of 89%. Moreover, for VEGF, the values were 91% of specificity, 78% of sensitivity, positive predictive value of 88% and negative predictive value of 83%.
AST to platelet ratio index (APRI)
This method uses two simple laboratory parameters, aspartate aminotransferase (AST) and platelet count. This rate is calculated as follows: APRI = (AST/upper limit x 100)/platelet count. As fibrosis progresses, portal hypertension, splenic sequestration and increased injury of platelets occurs, conferring a decrease in their levels. In addition, liver fibrosis is associated with decreased production of thrombopoietin, performed by hepatocytes. Furthermore, AST increases in liver fibrosis, since its clearance becomes reduced and its release by mitochondria increases due to hepatic injury, as reported by Shin et al.(5)
A differential of the study by Kruger et al.(6) in the year 2011 consists in the fact that the referred study was validated in patients whose liver disease is NAFLD, including its progress, such as NASH, which can evolve to fibrosis. It is usually validated in patients with chronic hepatitis C. As liver fibrosis progressed, APRI increased. In the group without fibrosis and with mild fibrosis, APRI presented a value of 0.7, whereas in the advanced fibrosis group the value increased to 1.54.
This method was also validated by Chang et al.(7) In this study, APRI was compared with another non-invasive method, the transient elastography (imaging method, which is not the focus of this review), that measures liver rigidity, attributing higher rigidity to fibrosis progression. Although the elastography proved being better than the blood test, the values found for APRI were promising.
In this study, patients with diverse chronic liver diseases were selected, such as: carriers of viral hepatitis; NASH; autoimmune hepatitis; primary biliary cirrhosis and elevated transaminases. It also included a group with unknown liver disease and undergoing liver surgery and one group of patients with cirrhosis waiting for resection of the carcinoma. APRI >1.50 was able to identify the presence/absence of significant fibrosis in 59.1% of the patients. This was the cut point chosen as optimal. Similarly, APRI >2.00 was able to identify the presence of cirrhosis in 80.9% of the patients.
It should be noticed that the accuracy of APRI can be affected by conditions such as hemolysis, cholestasis, and elevated transaminase levels.
AST to platelet ratio index (APRI)
This method uses two simple laboratory parameters, aspartate aminotransferase (AST) and platelet count. This rate is calculated as follows: APRI = (AST/upper limit x 100)/platelet count. As fibrosis progresses, portal hypertension, splenic sequestration and increased injury of platelets occurs, conferring a decrease in their levels. In addition, liver fibrosis is associated with decreased production of thrombopoietin, performed by hepatocytes. Furthermore, AST increases in liver fibrosis, since its clearance becomes reduced and its release by mitochondria increases due to hepatic injury, as reported by Shin et al.(5)
A differential of the study by Kruger et al.(6) in the year 2011 consists in the fact that the referred study was validated in patients whose liver disease is NAFLD, including its progress, such as NASH, which can evolve to fibrosis. It is usually validated in patients with chronic hepatitis C. As liver fibrosis progressed, APRI increased. In the group without fibrosis and with mild fibrosis, APRI presented a value of 0.7, whereas in the advanced fibrosis group the value increased to 1.54.
This method was also validated by Chang et al.(7) In this study, APRI was compared with another non-invasive method, the transient elastography (imaging method, which is not the focus of this review), that measures liver rigidity, attributing higher rigidity to fibrosis progression. Although the elastography proved being better than the blood test, the values found for APRI were promising.
In this study, patients with diverse chronic liver diseases were selected, such as: carriers of viral hepatitis; NASH; autoimmune hepatitis; primary biliary cirrhosis and elevated transaminases. It also included a group with unknown liver disease and undergoing liver surgery and one group of patients with cirrhosis waiting for resection of the carcinoma. APRI >1.50 was able to identify the presence/absence of significant fibrosis in 59.1% of the patients. This was the cut point chosen as optimal. Similarly, APRI >2.00 was able to identify the presence of cirrhosis in 80.9% of the patients.
It should be noticed that the accuracy of APRI can be affected by conditions such as hemolysis, cholestasis, and elevated transaminase levels.
FIB-4
The study of Lin et al.(8) evaluated APRI and FIB-4 separately and compared their accuracies. APRI has been already describe in this article. FIB-4 is an easily calculated index and includes AST, platelet count, age, and alanine aminotransferase (ALT). The method for calculating FIB-4 is shown below: FIB-4 = age (in years) x AST (U / L) / (platelet count (109 / L) x [ALT (U / L) 1/2 ]. This parameter has been validated in patients with chronic hepatitis B. The evaluation of said markers in patients carrying this disease is very important, because at least prior to the year of its publication, there were little data on the applicability of APRI and FIB-4 on these patients. For APRI an area under the receiver operating characteristics (AUROC) of 0.704 (in significant fibrosis) were shown. For FIB-4, the AUROC was 0.769. Comparing the accuracy of these two methods for cirrhosis, better results were found in the detection of cirrhosis than in significant fibrosis (AUROC of 0.706 in APRI and 0.869 in FIB-4). FIB-4 may have shown better results than APRI in both significant fibrosis and cirrhosis because it involves additional parameters related to liver fibrosis, such as age and ALT levels.
Coopscore
In the study by Taibi et al.,(1) this method has been validated in patients with chronic hepatitis C, comprising five parameters: alpha-2-macroglobulin, apolipoprotein-A1, AST, collagen IV and osteoprotegerin (OPG). The calculation model of the score is as follows: Coopscore = 1/(1 + exp (-) – 1.856 + 1.216 x lnA2M (mg/L) + 0.007 x C-IV (mg/L) – 1.047 x ApoA1/L) + 0.451 x ln OPG (mg/L) + 0.005 x AST (IU/L) + 0.005 x age + 0.231 x sex (M = 1, F = 0))).
Alpha-2-macrogolobulin is capable of inactivating various proteinases, inhibiting fibrolytic activity. In fibrotic liver, the increase of alpha-2-macroglobulin, with consequent inhibition of MMP and decrease of fibrosis degradation, favors the maintenance of this process, according to Ho et al.(9) In the case of apolipoprotein A1, Naveau et al.(10) describes that this analyte is present at lower levels in liver fibrosis. It is explained by the fact that apolipoprotein A1 attaches to extracellular matrix, in addition to decreasing the transcription of its RNA. Collagen IV has high levels in fibrosis, as well as OPG, a protein that regulates bone mass (altered during liver disease). Through an algorithm that combines the parameters presented previously, Coopscore was created.
The differential of the study by Taibi et al.(1) was the validation of Coopscore in patients coinfected with HBV and HIV. It is estimated that 5% to 10% of patients infected with HIV are also infected with HBV. Since HIV accelerates the consequences caused by HBV (progression from fibrosis to cirrhosis and increased incidence of HCC), the prognosis is poor.
The results showed that Coopscore increases as fibrosis levels increase. Furthermore, Obuchowski’s calculation revealed that the diagnostic performance of Coopscore was higher (0.774), compared to other patented tests, such as Fibrometer (0.668), Hepascore (0.690) and Zeng score (0.704, P <0.05), although they did not show differences between Coopscore and Fibrotest (0.734, P = 0.17). Despite this method has been shown promissory, it should be clarified that in certain cases, OPG levels may be altered, as in HIV infections associated with atherosclerosis and during bone remodeling, which could modify the results of Coopscore. In addition, the use of some antiretrovirals, so-called d-drugs (dideoxynucleotide analogues) and selective protease inhibitors are some other factors that may affect the levels of OPG and collagen IV.
Hepascore
This method consists of an algorithm that combines alpha-2 macroglobulin, hyaluronic acid, TIMP-1 and MMP-2. All these molecules are closely involved in the pathophysiology of liver fibrosis (their involvement in the pathophysiology of liver fibrosis has been reported in previous paragraphs). In addition to analytes, this method includes the age and sex of the patient as variables.
In this study, performed by Adams et al.,(11) an AUC of 0.820 was obtained for detection of significant fibrosis. For advanced fibrosis, the value was 0.903 and for cirrhosis 0.891 in the test validation group. Also, the raise in Hepascore values were proportional to increased fibrosis levels.
Various tests
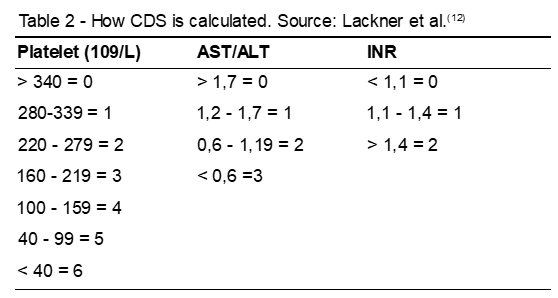
Lackner et al.(12) analyzed the accuracy of AST/ALT, cirrhosis discriminant score (CDS), age/platelet index (AP), Pohl score, APRI and platelet counts. The methods for obtaining some of these parameters are described below. The others were further explained previously.
CDS is obtained as follows: platelet count (x109/L), AST/ALT rate, and international normalized rate (INR). From the individual obtained values, the sum is made to obtain the final scores of CDS (Table 2).
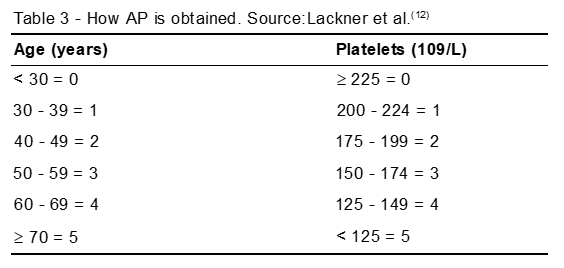
The AP is the sum of the scores obtained in age and platelet counts (Table 3).
In Pohl score: it is positive when AST / ALT ratio ³1 and platelet count <150 x 109 / L.
Liver fibrosis showed weak correlation values with ALT (M = 0.58), INR (M = 0.58), AST / ALT (M = 0.25) and CDS (M 0.46), while it was moderately related with AST (M 0.52), platelet count (M 0.48), AP index (M 0.52) and APRI (M 0.61). Subsequently, AUROC of platelet count, AP index and APRI were created. Among these, APRI presented the best prediction for fibrosis in the F3-F6 stages, of significant fibrosis (AUROC of 0.80) (according to the Ishak score). In the F5-F6 stages of cirrhosis, the best diagnostic power was given by the AP index (AUROC of 0.91).
The methods below are more related to experimental diagnosis.
Plasma free choline
The study by Imajo et al.(13) validated this method in patients with NASH. In animal models of NASH, researchers use mice with methionine and choline deficiency. Mice maintained on this diet are characterized by macrovesicular steatosis, hepatocellular death, inflammation, oxidative stress and fibrosis. These findings are similar to those seen in humans with NASH.
Choline deficiency appears to be associated with the mechanism responsible for the development of the pathogenesis, but this has not yet been fully elucidated. What is known is that choline is essential for the functioning of cells. Most of it is absorbed by small intestine and metabolized in liver. In addition, it is a precursor of phosphatidylcholine – the major component of very low density lipoprotein (VLDL). However, in this study, the finding was different from the animal models. In it, plasma choline levels have been shown to increase in NASH cases, compared with simple steatosis. One hypothesis that attempts to explain this controversial fact is that microsomal triglyceride transfer protein (MTTP) has reduced activity in NASH, and in fact this has been shown in previous studies. Therefore, free choline levels are increased in plasma.
Choline deficiency appears to be associated with the mechanism responsible for the development of the pathogenesis, but this has not been fully elucidated yet. It is known that choline is essential for the functioning of cells. Most of it is absorbed by the small intestine and metabolized in the liver. In addition, it is a precursor of phosphatidylcholine – the major component of very low density lipoprotein (VLDL). However, in this study, the finding was different from the animal models, because plasma choline levels appeared to increase in NASH cases, compared to simple steatosis. One hypothesis that attempts to explain this controversial fact is that microsomal triglyceride transfer protein (MTTP) has reduced activity in NASH, and in fact this has been elucidated in previous studies. As consequence, an increase in plasma free choline levels occurs.
Plasma free choline levels in NASH patients demonstrated to be significantly higher when compared to healthy patients (mean, 0.16 mg/dL, ranging from 0.10 to 0.25 mg/dL and a mean of 0.09 mg/dL, ranging from 0.07 to 0.12 mg/dL, respectively). Plasma free choline levels were also positively correlated with the degree of steatosis and fibrosis, but not with lobular inflammation and balloonization of hepatocytes (histological features present in NASH). Moreover, this variable does not change between different ages, genders, body mass indexes, levels of cholesterol, triacylglycerols, hyaluronic acid or presence/absence of obesity, diabetes, dyslipidemia or hypertension – which ends up being advantageous. On the other hand, its disadvantage is the specificity for patients with NAFLD and its progression (including NASH), since it has been shown that said analyte does not increase in other chronic liver diseases.
Serum N-glycomic markers
Along with plasma cells, hepatocytes are major sources of serum glycoproteins, since the liver is involved in clearance of proteins with aberrant glycosylation. Therefore, it is assumed that hepatic disorders may cause alterations in serum glycoprotein profile. Based on this idea, a study by Qu et al.,(14) has been conducted. Participants with HBV were divided in two groups: one with mild fibrosis and another with advanced fibrosis. Through DNA sequencer-aided fluorophore assisted carbohydrate electrophoresis technique (DSA-FACE), serum glycoproteins of these patients were separated and characterized by different peak patterns.
This technique is used to diagnose cirrhosis and hepatocellular carcinoma through the emerging of specific peak patterns. In cirrhosis, peak 7 is elevated while 8 is decreased, compared with healthy patients. Thus, the log derived from these peaks log(peak 7/peak 8) can distinguish cirrhotic patients. The intention was to discover some specific pattern to differentiate levels of fibrosis.
Results showed that peaks 1, 2, 3, 4 and 10 were significantly elevated, whereas 5, 8 and 11 decreased in advanced fibrosis when compared with the mild fibrosis group. Several peaks were obtained, so it was necessary to identify those that would be the best to fit fibrosis levels. Then, various receiver operating characteristic (ROC) curves were used to determine the role of different peaks. In this study, the best result was log(peak 2/peak 8). From this result, specificities, sensitivities, positive and negative predictive values and accuracy of different liver fibrosis stages were compared. Using log(peak2/peak8), good predictors were obtained for advanced fibrosis, but not for early and moderate stages.
Combinations of log(peak 2/peak 8) with biochemical methods already known were made, such as pro-collagen type III propeptide (PIIINP), collagen IV, HA, laminin, collagenases and inhibitors of collagenases. Among these, HA showed best correlation with different stages of fibrosis, although it did not improve AUROC with log(peak 2/peak 8) for diagnosis of significant, advanced fibrosis and cirrhosis.
Therefore, serum glycomic profile, although isolated, can predict well the presence of liver fibrosis, being more discriminant in significant fibrosis.
Serum connective tissue growth factor (CTGF)
CTGF is a member of the CYR61-CTGF-NOV 2 glycoprotein superfamily, a family of cysteine-rich glycoproteins, which is related to the pathogenesis of liver fibrosis. It is suggested that the referred protein is an amplifier of TGF-b, a pro-fibrogenic cytokine. Although its mechanism of action is not fully elucidated, it probably strengthens the binding of TGF-b1 to its receptor.
The study of Gressner et al.(15) showed that serum concentrations of CTGF were significantly elevated in HBV infected patients at the advanced fibrosis stage (stages 3 and 4, 36.9 ± 19.7 ng/mL), cirrhosis (43.6 ± 48, 5 ng/mL) and hepatocellular carcinoma (38.5 ng/mL) than in control 7.7 ± 10.7 ng/mL). However, this method was not able to discriminate well between healthy and fibrosis patients in the most early to moderate stages (stages 1 and 2, 20.3 ± 8.8 ng/mL).
Wisteria floribunda agglutinin-positive Mac-2 binding protein (WFA+ – M2BP)
WFA + – M2BP protein has already been reported as a glycobiomarker for liver fibrosis. It is secreted primarily by hepatocytes. WFA + – M2BP mediates cell proliferation, angiogenesis, and is also involved in the increase of cell adhesion to extracellular matrix. Their serum levels increase as fibrosis progresses. In the study by Ura et al., (16) hepatitis C was evaluated, wherein the identification of significant fibrosis is crucial, since patients with fibrosis at this stage have a higher chance of rapidly progressing to hepatocellular carcinoma. Thus, if this stage of significant fibrosis is reached, these individuals should be promptly medicated, in order to suppress the progression of the disease. The advantages of using this method are its liver specificity, high diagnostic accuracy and good reproducibility. For ? F2 (Metavir score), WFA + – M2BP quantification revealed sensitivity of 83.6, specificity of 73.6, positive predictive value of 65.7 and negative predictive value of 88.2. For ³ F3, sensitivity was 78.4, specificity 64.2, positive predictive value 42.6 and negative predictive value 89.7.
Discussion
More than 50% of patients with cirrhosis die within the first 17 years after diagnosis, mostly with hepatocellular carcinoma. Therefore, attenuation of the fibrogenic process is important to decrease morbidity. Due to its invasive nature and serious analytical limitations, histological evaluation is not recommended for the purpose of diagnosis and monitoring, according to Taibi et al.(1) Additionally, the prognosis is also important because, in hepatitis C cases, e.g., the infection depends on the current fibrosis stage and the progression rate of the disease (fibrogenic rate). Therefore, the sooner the diagnosis is made, the better for the clinician, being able to propose the treatment, and even better for the patient, that would return to its activities without great harm to its health. However, the course of chronic liver disease can take decades to generate symptoms that enable proper investigation and diagnosis. Thus, the patient often receives diagnosis when in terminal phase and may require liver transplantation. At a terminal stage, treatment may not be effective.
Ideally, hepatic monitoring should be performed, especially when the patient is exposed to risk factors, such as excessive alcohol consumption, presence of hepatic steatosis, etc. However, it is not feasible to perform biopsies at short intervals, since this method presents risks to the patient because it is highly invasive, expensive, depends on trained professionals to obtain a significant sample and the method itself has disagreements, because the reading is done by human beings, undergoing individual subjectivity. In this sense, the development of non-invasive diagnostic methods is of great importance. In addition, benefits of these methods would also be seen during the monitoring of chronic liver diseases, since they could dispense repeated biopsies and consequent discomfort for the patient.
From the analyzed studies, it is noted that hepatitis C is the chronic liver disease widely chosen for study of diagnostic methods. However, it is important to note that fibrosis is a common consequence of dozens of liver diseases. Privileging a disease in the study ends up impairing the analysis of quality of these markers, after all, not necessarily all analytes will present similar behaviors in all situations.
Unlike other methods, which appeared only once in the present study, APRI appeared three times. Probably, due to platelets and AST being analytes commonly evaluated in laboratory routine. Some of these studies validated the method in NAFLD and hepatitis B. These are promising ways to evaluate fibrosis arising from other liver diseases besides hepatitis C. However, this method is more reliable in cases where liver fibrosis is in advanced level (cirrhosis), it may be useful in cases where advanced fibrosis is confirmed, so a non-invasive method can be applied for monitoring, since fibrotic patients are usually evaluated with constancy.
FIB-4 is another very simple method to use. It takes in account age, AST, ALT and platelet count. In addition, the study by Lin et al.(8) showed that, in comparison with APRI, FIB-4 can be more accurate, because it considers other parameters than those in APRI. Homocysteine and VEGF are analytes that also have their levels checked regularly in clinical analysis laboratories, but not as often as AST, ALT, and platelets. In the study by Polyzos et al.(3) it has been seen that homocysteine levels are inversely related to fibrosis progression. The disadvantage is that it is closely related to the metabolic syndrome, so its use is only seen in cases where fibrosis is related to NAFLD. The right decision would be analyze it in other hepatic diseases, allowing possible discoveries of correlation with other illness.
Other tests are more related to scientific research field. We can cite N-glycomic markers, serum CTGF, free plasma choline, and WFA + – M2BP. However, this represents a start for the discovery of new and promising diagnoses of liver fibrosis. It is worth noticing that none of the four prior said tests proved to be precise for diagnosis of fibrosis at earlier stages. Among them, choline has the advantages of being stable in several secondary pathologies, although it is only useful in cases where fibrosis comes from NAFLD.
HA is a direct marker of liver fibrosis, that reflects its metabolism. In the research conducted by Toson et al.(4) it has been shown that HA is a good predictor of liver fibrosis. In addition to direct markers, there are algorithms that can use combinations of direct and indirect markers (indirect ones reflect changes in liver function as a result of fibrosis) and can be combined with patient variables such as sex and age. This group includes Coopscore and Hepascore. Both have their values directly related to progression of fibrosis. In addition, Coopscore study had the differential of being analyzed in patients HBV-HIV coinfected, opening doors to works that could compare the accuracy of diagnostic methods in mono and coinfected patients.
It is possible to note that there is a variety of markers studied in different liver diseases, which use several scales for their classification. In addition, the groups of patients analyzed were heterogeneous. This means that we must always keep in mind that studies conducted in sites with diverse incidence and prevalence of a particular chronic liver disease may present changes in test sensitivity and specificity.
The ideal marker should be able to accurately access fibrosis severity with reliability, reproducibility and monitoring of fibrosis. Also, it should be available in most, if not all, hospitals.
Conclusion
The fact of a marker being validated in a specific disease does not exclude the possibility of using it in other chronic hepatic etiologies. In order to know which markers can be used and in which diseases, studies that contemplate a greater number of diseases (including concomitants) using the same analyte or a set of them are necessary. With more advanced research, summed to molecular biology, new markers can be discovered, studied and, therefore, its reference values applied to a range of chronic liver diseases.
The research on non-invasive diagnostic and monitoring methods for liver fibrosis that are cheaper and easier to access in different regions of the world is notable. Despite the efforts, there are many barriers to be faced, such as the difficulty of standardization in different groups studied and variations of the analytes in various chronic liver diseases. However, the existence of the methods discussed in this study is a path for a promising future in the non-invasive diagnosis of fibrosis.
Resumo
Introdução: A fibrose hepática é uma das consequências graves de doenças hepáticas crônicas. Seu diagnóstico precoce é de suma importância para uma tomada rápida de decisão para tratamento e prognóstico. Seu diagnóstico e monitoramento costuma ser realizado por biópsias hepáticas, método custoso, com grandes chances de complicações e interpretação de resultados de forma subjetiva. Objetivos: A pesquisa para o desenvolvimento de métodos não invasivos é grande. O objetivo deste estudo é mostrar e avaliar alguns desses métodos. Materiais e métodos: Foram analisados artigos de validação em quatro bases de dados (Medline, Cochrane, LILACS e SciELO). Os termos empregados foram: (liver fibrosis) AND (non-invasive diagnosis). Todos os trabalhos sucederam aos métodos inclusão e exclusão. Resultados: Alguns métodos se mostraram promissores, sendo grande parte utilizados no diagnóstico mais tardio da fibrose hepática. Discussão: Muitos métodos foram e estão sendo validados, alguns se mostrando promissores, embora necessitem de futuros refinamentos em suas avaliações, como diversificação da população estudada e avaliação em diversas doenças hepáticas crônicas. Conclusões: Ainda existem muitas dificuldades para criação e padronização dos métodos analíticos. Todavia, a existência e a validação são passos importantes para um futuro favorável no diagnóstico de fibrose.
Palavras-chave
Cirrose hepática; hepatopatias; biópsia
REFERÊNCIAS
- Taibi L, Boyd A, Bosselut N, Bottero J, Guechot J, Lacombe K, et al. Diagnostic accuracy of the Coopscore((c)) to predict liver fibrosis in human immunodeficiency virus/hepatitis B virus co-infection. Ann Clin Biochem. 2018;55(2):236-43. doi: 10.1177/0004563217707979.
- Ichikawa Y, Joshita S, Umemura T, Shobugawa Y, Usami Y, Shibata S, et al. Serum Wisteria floribunda agglutinin-positive human Mac-2 binding protein may predict liver fibrosis and progression to hepatocellular carcinoma in patients with chronic hepatitis B virus infection. Hepatol Res. 2017;47(2):226-33. doi: 10.1111/hepr.12712.
- Polyzos SA, Kountouras J, Patsiaoura K, Katsiki E, Zafeiriadou E, Deretzi G, et al. Serum homocysteine levels in patients with nonalcoholic fatty liver disease. Ann Hepatol. 2012;11(1):68-76.
- Toson EA, Shiha GE, Abdelgaleel AE. Fibrogenic/Angiogenic Linker for Non-Invasive Assessment of Hepatic Fibrosis Staging in Chronic Hepatitis C Among Egyptian Patients. Ann Hepatol. 2017; 16(6): 862-73. doi: 10.5604/01.3001.0010.5276.
- Shin WG, Park SH, Jang MK, Hahn TH, Kim JB, Lee MS, et al. Aspartate aminotransferase to platelet ratio index (APRI) can predict liver fibrosis in chronic hepatitis B. Dig Liver Dis. 2008;40 (4):267-74.
- Kruger FC, Daniels CR, Kidd M, Swart G, Brundyn K, van Rensburg C, et al. APRI: a simple bedside marker for advanced fibrosis that can avoid liver biopsy in patients with NAFLD/NASH. S Afr Med J. 2011;101(7):477-80.
- Chang PE, Lui HF, Chau YP, Lim KH, Yap WM, Tan CK, et al. Prospective evaluation of transient elastography for the diagnosis of hepatic fibrosis in Asians:comparison with liver biopsy and aspartate transaminase platelet ratio index. Aliment Pharmacol Ther. 2008;28(1):51-61. doi: 10.1111/j.1365-2036.2008.03711.x.
- Lin CL, Liu CH, Wang CC, Liang CC, Su TH, Liu CJ, et al. Serum Biomarkers Predictive of Significant Fibrosis and Cirrhosis in Chronic Hepatitis B. J Clin Gastroenterol. 2015;49(8):705-13. doi: 10.1097/MCG.0000000000000250.
- Ho AS, Cheng CC, Lee SC, Liu ML, Lee JY, Wang WM, et al. Novel biomarkers predict liver fibrosis in hepatitis C patients: alpha 2 macroglobulin, vitamin D binding protein and apolipoprotein AI. J Biomed Sci. 2010 Jul 15;17:58. doi: 10.1186/1423-0127-17-58.
- Naveau S, Raynard B, Ratziu V, Abella A, Imbert-Bismut F, Messous D, et al. Biomarkers for the prediction of liver fibrosis in patients with chronic alcoholic liver disease. Clin Gastroenterol Hepatol. 2005 Feb;3(2):167-74.
- Adams LA, Bulsara M, Rossi E, DeBoer B, Speers D, George J, et al. Hepascore: an accurate validated predictor of liver fibrosis in chronic hepatitis C infection. Clin Chem. 2005;51(10):1867-73.
- Lackner C, Struber G, Liegl B, Leibl S, Ofner P, Bankuti C, et al. Comparison and validation of simple non-invasive tests for prediction of fibrosis in chronic hepatitis C. Hepatology. 2005;41(6):1376-82.
- Imajo K, Fujita K, Yoneda M, Shinohara Y, Suzuki K, Mawatari H, et al. Plasma free choline is a novel non-invasive biomarker for early-stage non-alcoholic steatohepatitis: A multi-center validation study. Hepatol Res. 2012;42(8):757-66. doi: 10.1111/j.1872-034X.2012.00976.x.
- Qu Y, Gao CF, Zhou K, Zhao YP, Xu MY, Lu LG. Serum N-glycomic markers in combination with panels improves the diagnosis of chronic hepatitis B. Ann Hepatol. 2012;11(2):202-12.
- Gressner OA, Fang M, Li H, Lu LG, Gressner AM, Gao CF. Connective tissue growth factor (CTGF/CCN2) in serum is an indicator of fibrogenic progression and malignant transformation in patients with chronic hepatitis B infection. Clin Chim Acta. 2013; 421:126-31. doi: 10.1016/j.cca.2013.02.029.
- Ura K, Furusyo N, Ogawa E, Hayashi T, Mukae H, Shimizu M, et al. Serum WFA(+) -M2BP is a non-invasive liver fibrosis marker that can predict the efficacy of direct-acting anti-viral-based triple therapy for chronic hepatitis C. Aliment Pharmacol Ther. 2016;43 (1):114-24. doi: 10.1111/apt.13431.
Correspondência
Graciele Gonçalves Santos
Avenida Carlos Chagas Filho, 373, Cidade Universitária
Laboratório de Imunoparasitologia e Análises Toxicológicas (LIPAT)
Bloco A, 2º Andar, sala 017
Rio de Janeiro-RJ, Brasil
